1 / 5
 Evolve Medical
Evolve Medical











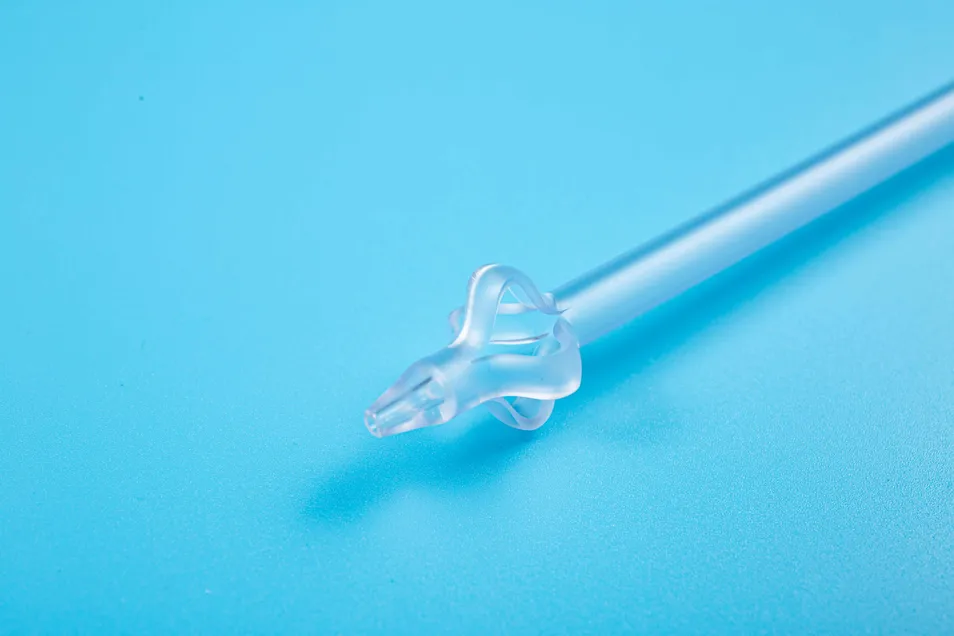
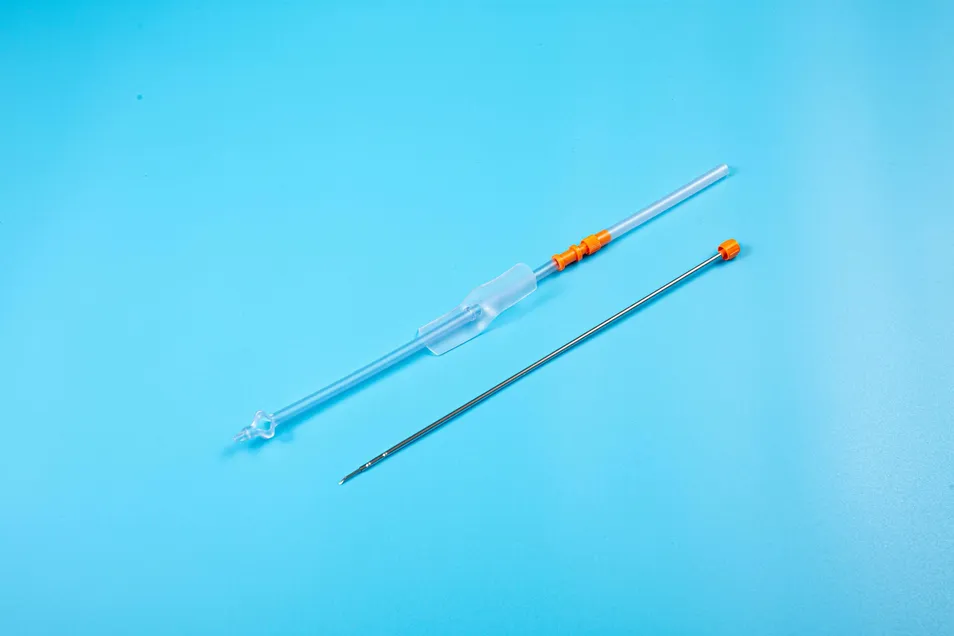
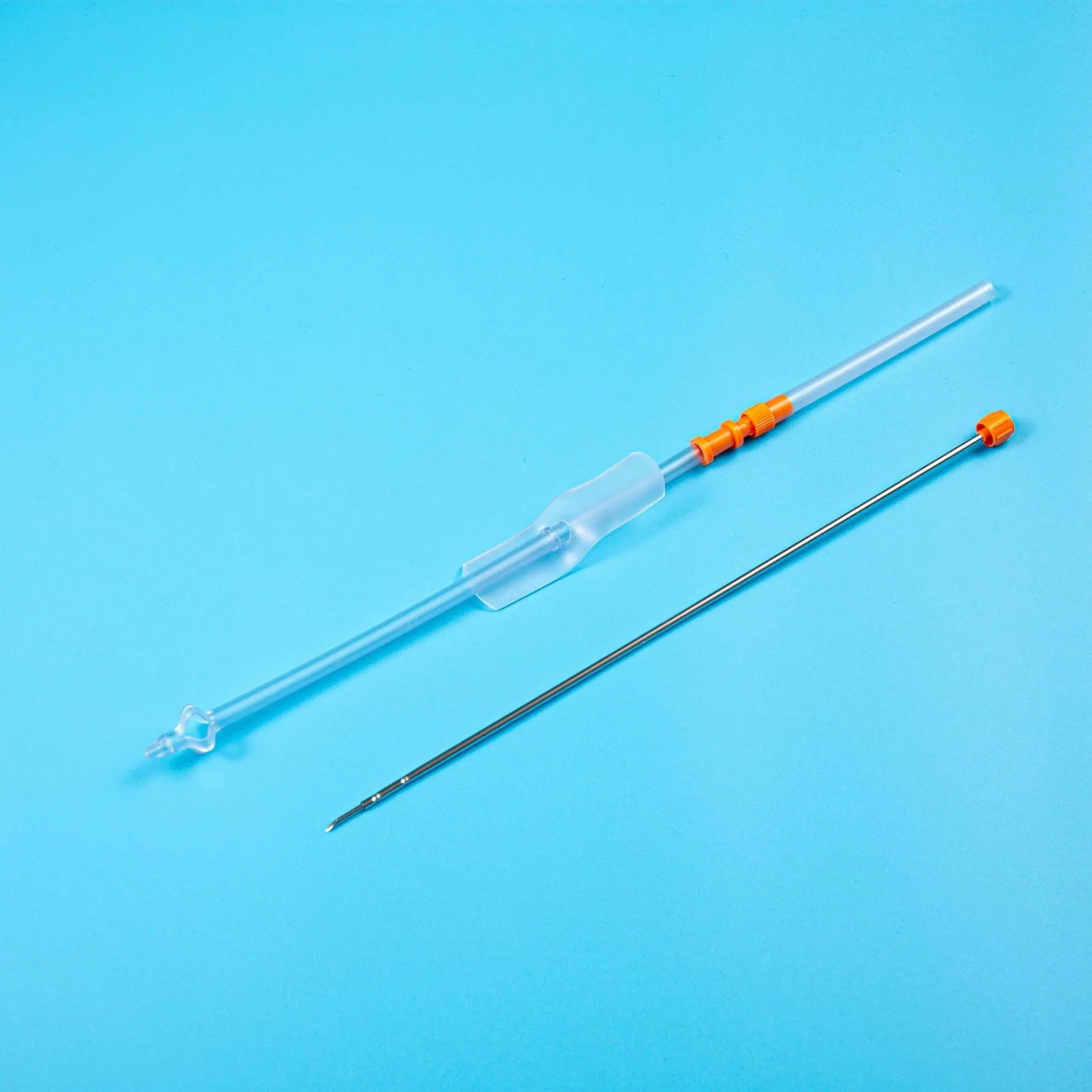
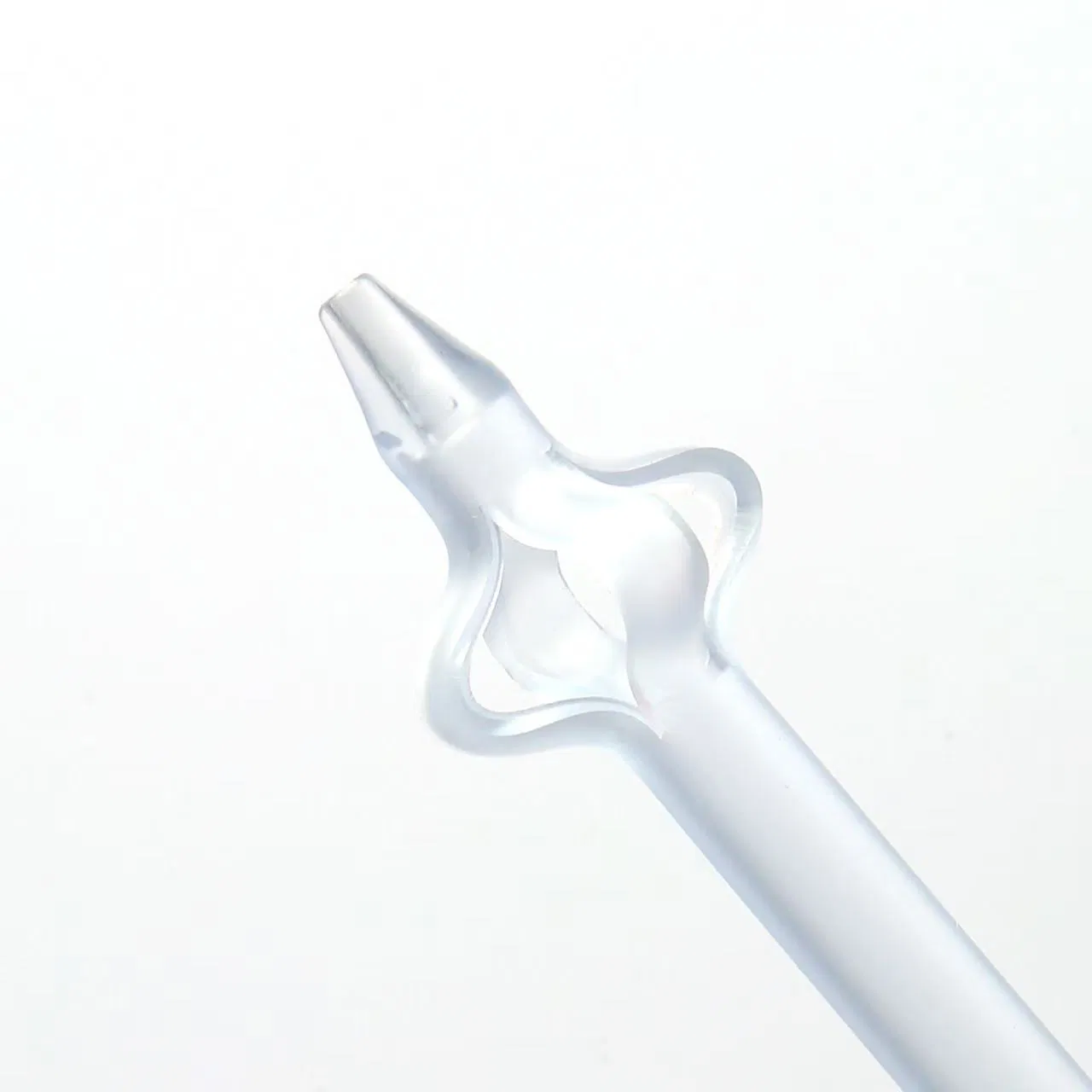
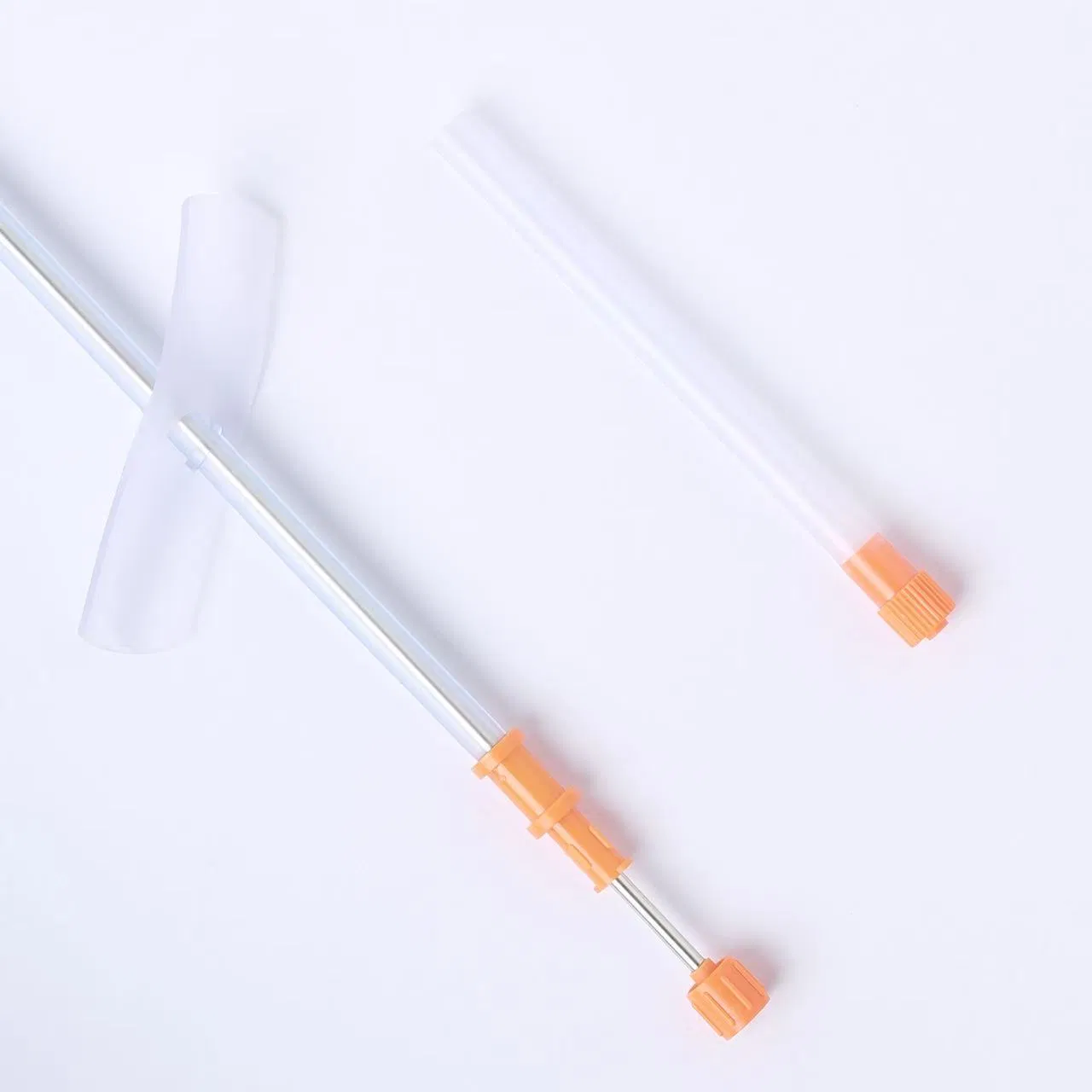
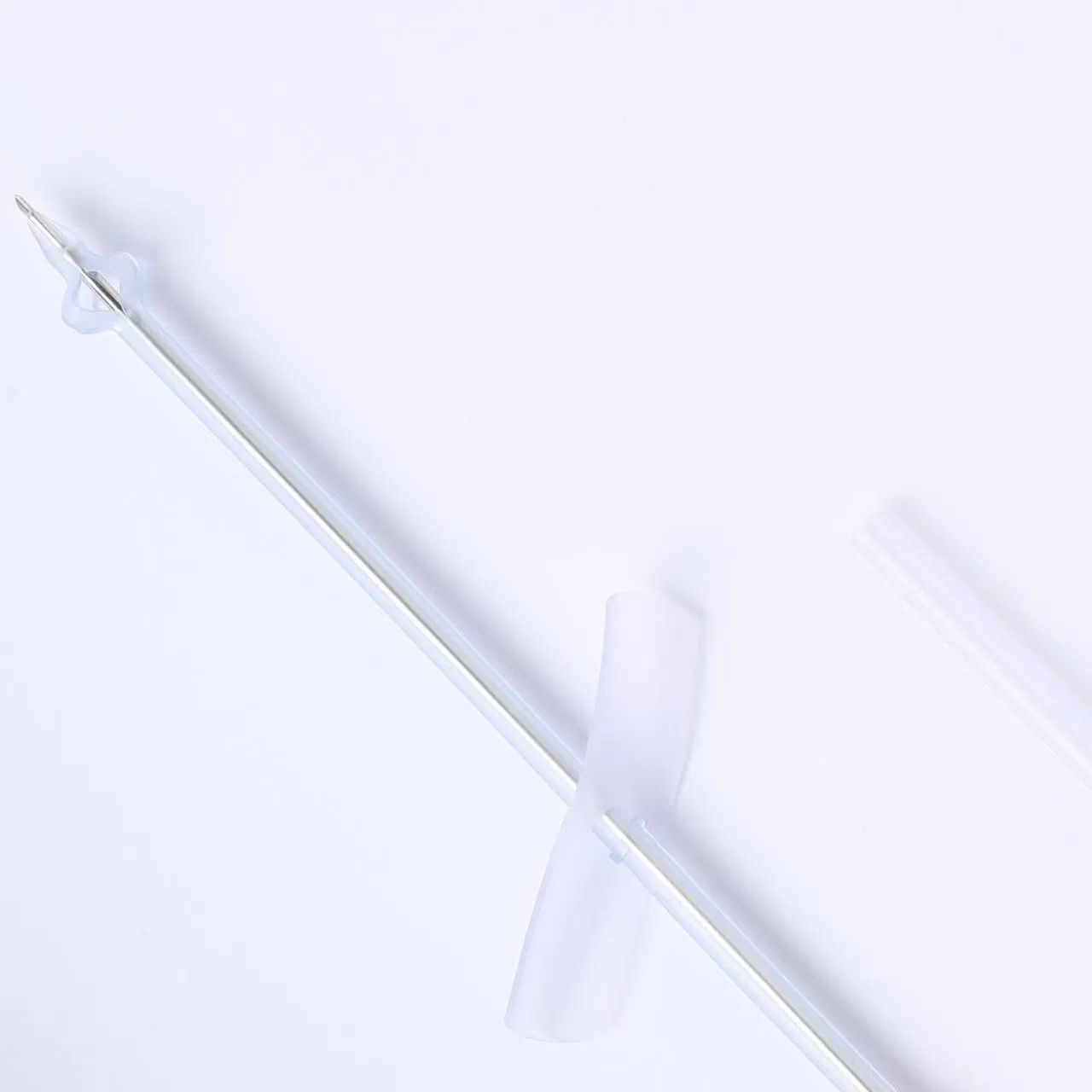
| Product Name | Disposable Sterile Suprapubic Cystostomy Catheter |
| Model NO. | Complete Set / CT-01-18 |
| Specifications | F14, F18, F24 |
| Length | 200mm |
| Material | Silicone-rubber |
| Sterilization | Ethylene Oxide (EOS) |
| Validity | 3 Years |
| Certification | ISO13485 |
| Classification | Class II |
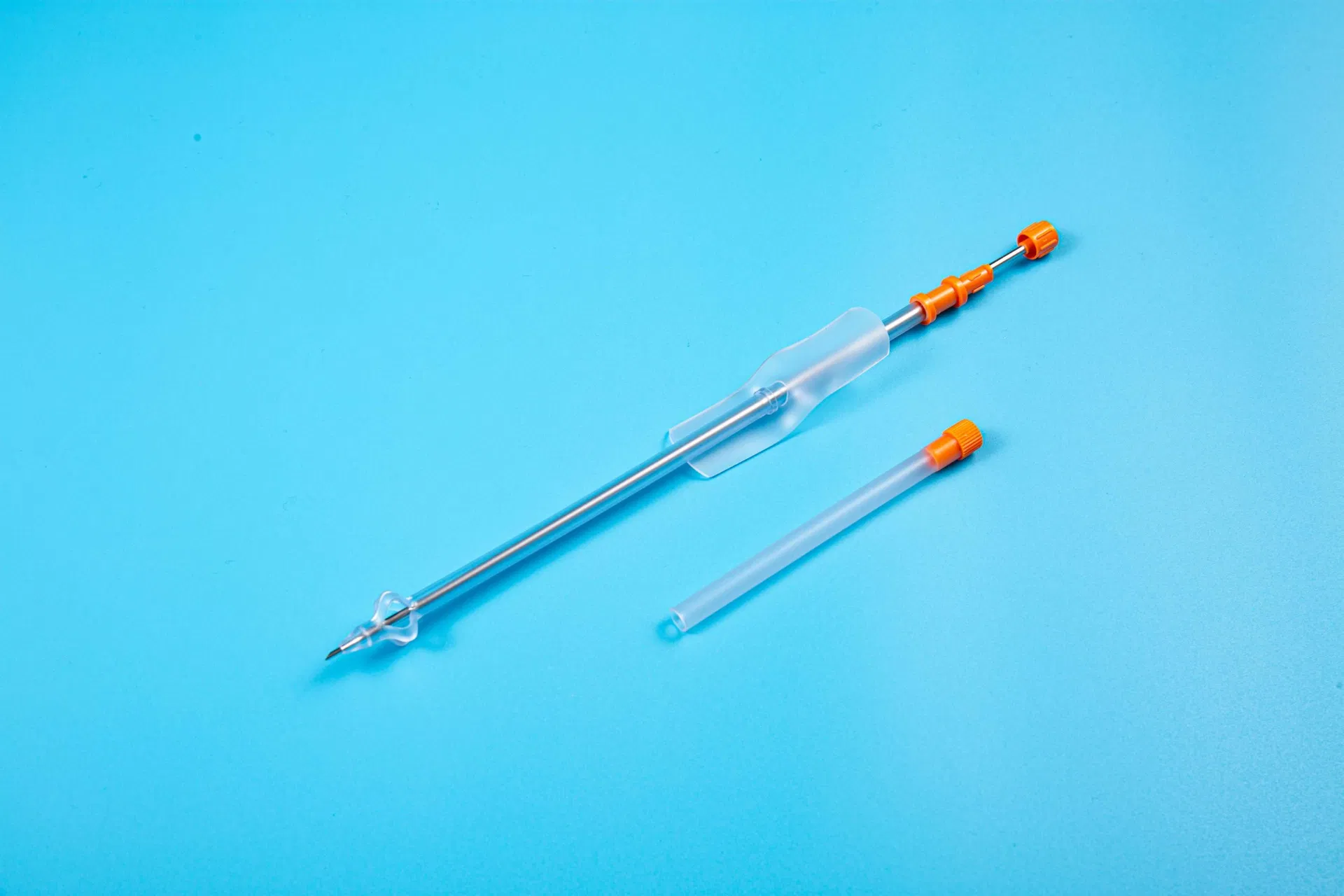
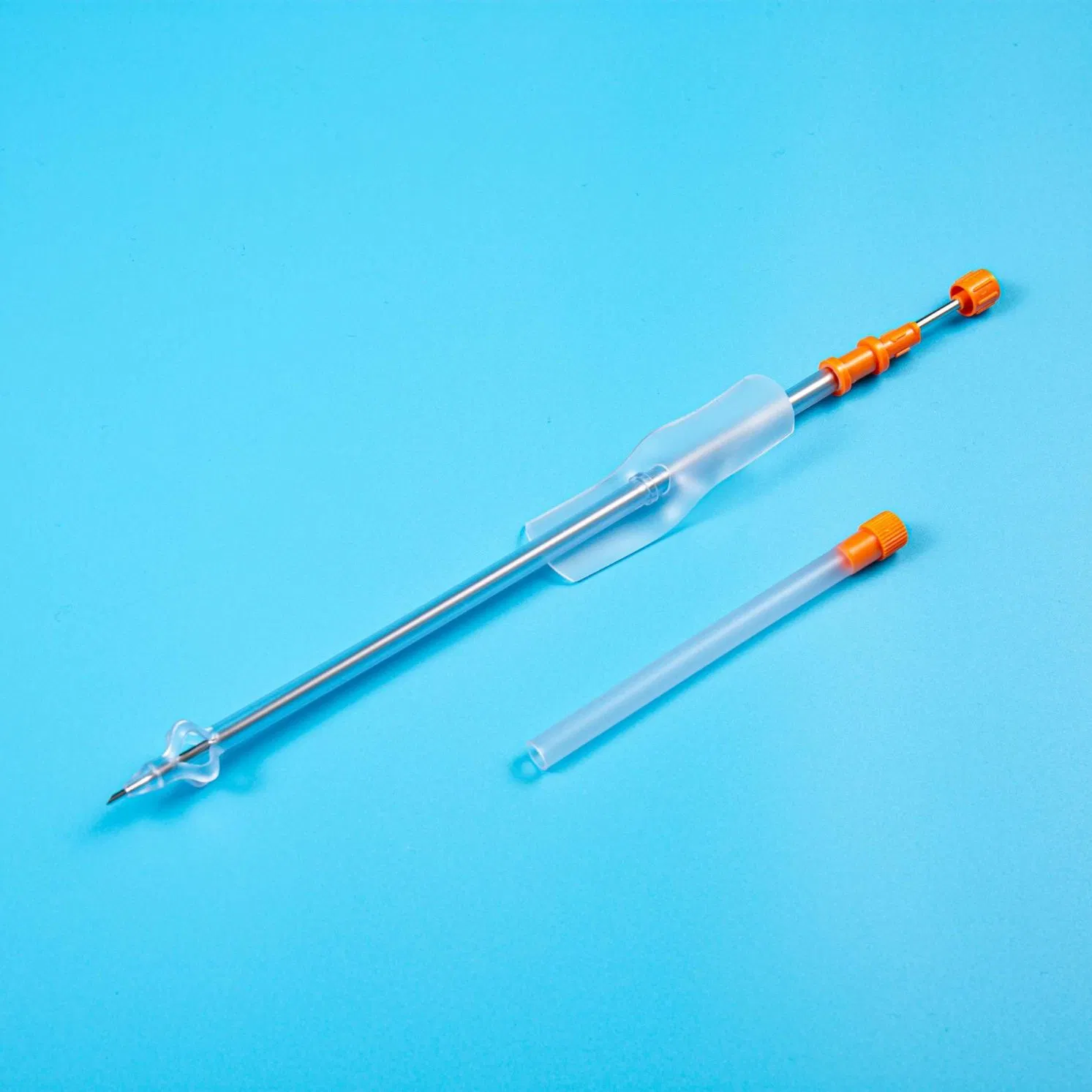
The product is applied for temporarily urinary diversion, suprapubic bladder puncture fistula, and effective drainage of urine.
Reduces the risk of urinary system infections while providing a reliable long-term drainage channel for patients with urinary retention.