




 Evolve Medical
Evolve Medical










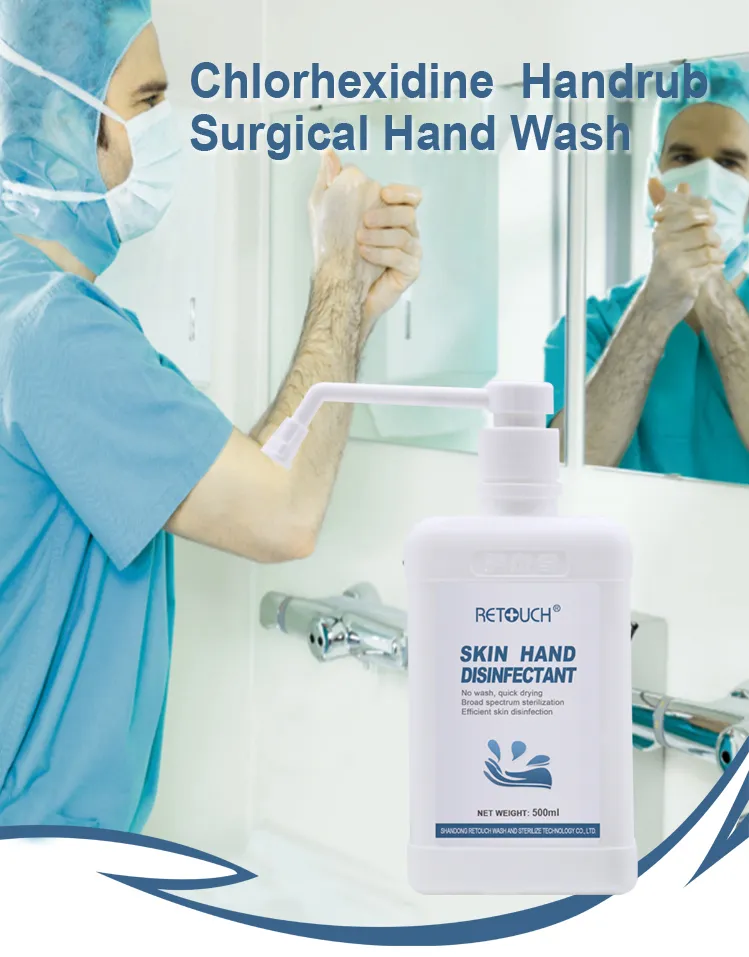
This disposable hand disinfectant is a disinfectant with Surgical chg gluconate and ethanol as the main active ingredient. The content of Chlorhexidine gluconate is 1% + 0.1% (W/V), and the content of ethanol is 70% + 5% (V/V).
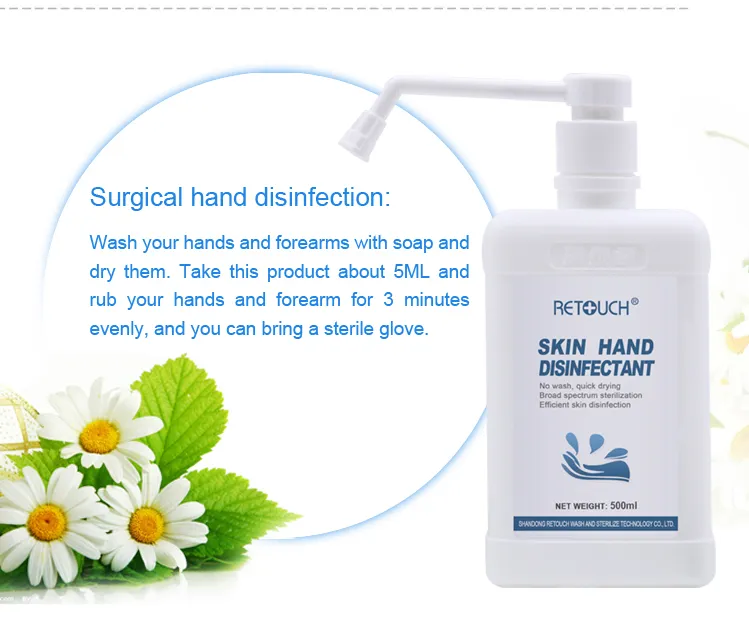
It is mainly suitable for hand disinfection, local skin disinfection before injection and infusion, and skin disinfection of surgical site.
| Active ingredient | Chlorhexidine gluconate and ethanol |
| Effective Content | Chlorhexidine gluconate 1% + 0.1% (W/V); ethanol 70% + 5% (V/V) |
| Application | Hand, Skin surface |
| Killing bacteria | Intestinal pathogenic bacteria; Pyogenic coccus; Pathogenic microzyme |
| Specifications | 50ml, 100ml, 500ml, 1L, Customized |
| Term of validity | 24 months |
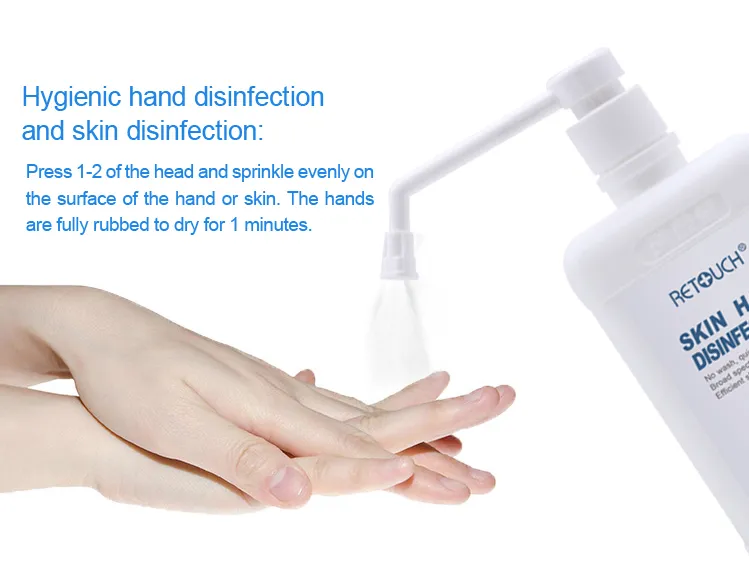
| Features | 1. Free from washing 2. Kill 99.999% of harmful bacteria 3. Quick disinfection |
| Attention | External use only; stay away from children; contains ethanol (avoid if allergic to alcohol). |


100ml, 500ml, 1gallon, 5L/barrel, customized according to requirements.




Founded in July 2001, our facility is a new and high-tech company specialized in research, development, manufacturing, and marketing promotion of hygiene disinfection products. It is a noted company in the domestic disinfection industry.
Sticking to the development tenet of 'invigorating the company with technology', we aim for 'leading a sterile new world' as development objectives, having obtained upward of 20 proprietary intellectual property rights and multiple patented technologies.

We have passed ISO9001:2008 quality management system certification. Products are exported to EU, Asian Pacific, and African countries. The company has established an independent laboratory testing center and technology lab equipped with advanced testing devices.
